COVID-19 Vaccine for 6 Months – 4 Years Coming Soon!

The wait is almost over! COVID Vaccines for under 5 have been approved and recommended by the CDC. Our first doses of the Pfizer vaccine should be arriving within days. Once we have vaccines and authorization from the Oregon Health Authority to administer them we will let you know!
EPPC doctors and staff eagerly anticipate the approval and subsequent delivery later this month (June) of COVID-19 vaccine for infants 6 months through 4 years of age. We are in the midst of planning vaccine clinics in late June and July, so that we can effectively and efficiently provide the vaccine to your child(ren). Though we cannot schedule appointments until the vaccine has actually arrived at our clinic, we encourage you to become informed and ready to vaccinate your infant/child with the COVID-19 vaccine. We believe the vaccine is safe and confidently recommend it.
- COVID-19 Vaccine for Infants, Toddlers and Young Children – What to Expect
- An emergency doctor and parent sorts through Covid vaccine options for kids under 5 – STAT
- About the COVID-19 Vaccine: Frequently Asked Questions – AAP
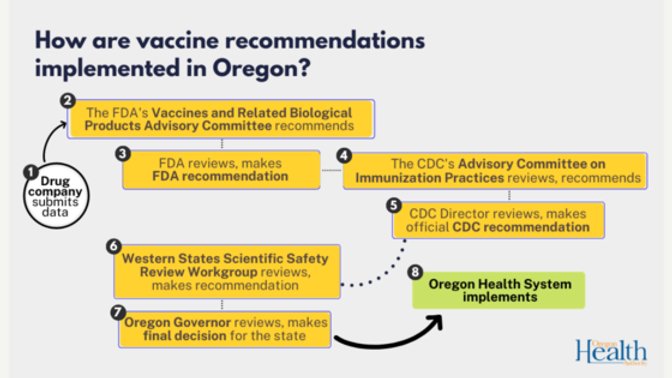
The CDC Advisory Committee on Immunization Practices meets Wednesday, June 15, and we could have vaccine doses early the week of June 20. Once we know how many doses we received, we will open up schedules and send out a broadcast message.
Both Pfizer and Moderna vaccines are expected to be approved.
- The Pfizer vaccine is a 3 dose series with 3 mcg doses.
- The Moderna is a 2 dose series with 25 mcg doses.
As of now we are expecting only Pfizer vaccine doses, as our order was placed weeks ago.
Booster Doses
Booster doses of COVID-19 have already been approved for children 5 years and older. We encourage the booster dose if at least 5 months have passed since your child’s second COVID-19 vaccine. Please call our office to schedule this booster dose if your child has met the timeline eligibility.

As always we welcome the opportunity to discuss any health matter, including vaccines, with you.
